|
The sum of the charges of the reactants equals the sum of the charges of the products.The sum of the mass numbers of the reactants equals the sum of the mass numbers of the products.Nuclear reactions also follow conservation laws, and they are balanced in two ways: A balanced nuclear reaction equation indicates that there is a rearrangement during a nuclear reaction, but of nucleons (subatomic particles within the atoms’ nuclei) rather than atoms. Balancing Nuclear ReactionsĪ balanced chemical reaction equation reflects the fact that during a chemical reaction, bonds break and form, and atoms are rearranged, but the total numbers of atoms of each element are conserved and do not change. Due to the much larger energy differences between nuclear energy shells, gamma rays emanating from a nucleus have energies that are typically millions of times larger than electromagnetic radiation emanating from electronic transitions. Gamma rays are a type of high energy electromagnetic radiation produced when a nucleus undergoes a transition from a higher to a lower energy state, similar to how a photon is produced by an electronic transition from a higher to a lower energy level. −1 0 e + +1 0 e ⟶ γ + γ −1 0 e + +1 0 e ⟶ γ + γĪs seen in the chapter discussing light and electromagnetic radiation, gamma rays compose short wavelength, high-energy electromagnetic radiation and are (much) more energetic than better-known X-rays that can behave as particles in the wave-particle duality sense. This works because, in general, the ion charge is not important in the balancing of nuclear equations. For example, an alpha particle is a helium nucleus (He) with a charge of +2 and a mass number of 4, so it is symbolized 2 4 He. The subscripts and superscripts are necessary for balancing nuclear equations, but are usually optional in other circumstances. Positrons ( +1 0 e, ( +1 0 e, also represented by the symbol +1 0 β ) +1 0 β ) are positively charged electrons (“anti-electrons”). Beta particles ( −1 0 β, ( −1 0 β, also represented by the symbol −1 0 e ) −1 0 e ) are high-energy electrons, and gamma rays are photons of very high-energy electromagnetic radiation. Alpha particles ( 2 4 He, ( 2 4 He, also represented by the symbol 2 4 α ) 2 4 α ) are high-energy helium nuclei. Protons ( 1 1 p, ( 1 1 p, also represented by the symbol 1 1 H ) 1 1 H ) and neutrons ( 0 1 n ) ( 0 1 n ) are the constituents of atomic nuclei, and have been described previously. The most common are protons, neutrons, alpha particles, beta particles, positrons, and gamma rays, as shown in Figure 21.4. Many entities can be involved in nuclear reactions. To describe a nuclear reaction, we use an equation that identifies the nuclides involved in the reaction, their mass numbers and atomic numbers, and the other particles involved in the reaction. Identify common particles and energies involved in nuclear reactionsĬhanges of nuclei that result in changes in their atomic numbers, mass numbers, or energy states are nuclear reactions.Each unique substance in the chemical reaction is sunder by a plus sign (+).By the end of this section, you will be able to: The reactants and products are sunders by arrow symbols. The input equation should be in the following formatĪ chemical equation is interpreted as the symbolic representation of the chemical reaction where the reactants are written on the left side and the products are written on the right side. 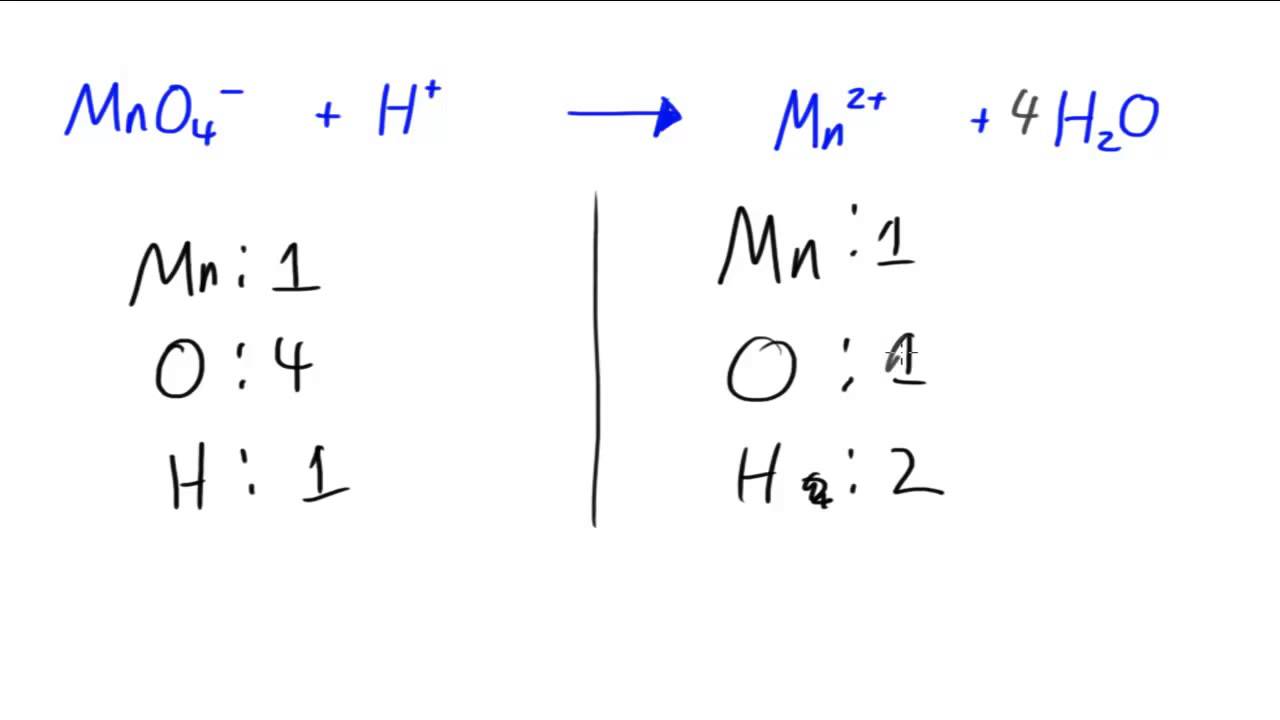
Method to use the Ionic net equation calculator is as follows:ġ: Enter the chemical equation in the “Enter the chemical equation” field.Ģ: Now click the button “Balance” to get the equalize equation.ģ: Finally, for the specified chemical equation, a window will pop with the output. How to use Net Ionic Equation Calculator? 
The balanced net ionic equation calculator tool makes the prediction quick and easier and displays the answer in a fraction of seconds.  
Net ionic and ionic equation calculator is a free online tool that shows the structure, equilibrium constant, balanced equation, substance properties with chemical formulas and names.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
